Understanding Photobiomodulation Therapy
A new energy-based treatment for inflammatory eyelid disease.
Craig Thomas, OD
GLOSSARY
- Bioenergetics is the scientific discipline that studies the production, distribution, and use of energy through living organisms.
- Cellular bioenergetics refers to biochemical reactions involved in energy metabolism, energy relationships, energy transformations, and transductions in living organisms.
- Cellular respiration describes the delivery of metabolites and oxygen to the mitochondria, oxidation of reduced metabolic fuels with passage of electrons to oxygen, and synthesis of chemical energy known as adenosine triphosphate.
Like many modern optometrists, I do not subscribe to the traditional classification of blepharitis (ie, anterior vs posterior blepharitis). In fact, I prefer to use the term inflammatory eyelid disease because I think it describes the condition more accurately.
More important than how we choose to classify or describe blepharitis is how we treat it. Optometrists now have two types of biostimulation devices to choose from when delivering photobiomodulation therapy (PBMT) to patients with inflammatory eyelid disease: intense pulsed light (IPL) and low-level light therapy (LLLT).
IPL is an energy-based technology that has been used for several years, especially in dermatology, and has recently been shown to be safe and effective for treating patients with meibomian gland dysfunction and reducing symptoms of dry eye disease.1
LLLT, on the other hand, despite having been around for more than 50 years, has only recently become available as an office-based device. Because IPL therapy has been covered in numerous articles and videos, the remainder of this article focuses solely on LLLT, a form of PBMT.
THE SCIENCE BEHIND LLLT
Photobiology and Photobiomodulation
Photobiology is the scientific discipline that studies the effects of light on living organisms and their biologic processes. The first law of photobiology states that photons of light must be absorbed by some molecule located within the cells to have a biological effect.
FitEyes Editor Note: The above statement seems too broad to me. Within the area of photochemistry as a subset of photobiology, the first law explains that in order for a photochemical reaction to happen, light has to be absorbed (by a molecule). That does not, as far as I can tell, mean there is a universal law that light must be absorbed by some molecule located within the cells to have any biological effect.
In LLLT, red or near-infrared light energy is used to modulate cellular bioenergetics and thereby increase energy production. LLLT interventions can also reduce inflammation by modulating immune system responses.2
The initial photon absorption event occurs in the cellular mitochondria, and the principal molecule that absorbs the light is an enzyme called cytochrome c oxidase (Cco).3
Cellular Respiration and Photoactivation
The enzymatic activity of Cco is known to reduce oxygen to water at the end of the mitochondrial respiratory chain, a necessary step in maintaining healthy cellular respiration.4
When cells are damaged by disease, injury, or aging, the mitochondria within the cells produce excess nitric oxide (NO), which can inhibit electron transport within the mitochondria and decrease mitochondrial membrane potential. Too much NO also shuts down the production of adenosine triphosphate (ATP) by competitively displacing oxygen from Cco. This displacement of oxygen also causes an overproduction of reactive oxygen species (ROS) and leads to oxidative stress. Untreated, the combined effects of abnormal cellular respiration can lead to decreased cell metabolism and premature cell death.5
Cellular photoactivation is achieved after absorption of the excitation energy. The leading hypothesis is that the energy transferred from the photon absorption breaks the chemical bond between NO and Cco that is inhibiting the mitochondrial electron transport chain.
Photodissociation with red light allows oxygen to replace the inhibitory NO from the binding site on the enzyme, which immediately reverses the inhibition of cellular respiration, thereby improving cell metabolism.2
Antiinflammatory Mechanisms
Although photon absorption by Cco is the first step in the intracellular photosignaling pathway, studies suggest that mitochondrial photoactivation initiates the release of secondary mediators such as cyclic adenosine monophosphate, NO, and ROS, which generate the molecules that initiate the signaling cascades that produce antiinflammatory effects.
The ability of secondary mediators to modulate calcium levels and activate transcription factors and signaling pathways is the proposed explanation for how a relatively brief exposure to red light can have long-lasting antiinflammatory results.2
In addition, a recent study of blepharitis in a rat model revealed that laser photobiomodulation treatments at 808 nm were effective in lowering the levels of inflammatory cytokines interleukin-1B and tumor necrosis factor alpha in the eyelids and the conjunctiva after 1 month of treatment.1
Antibiotic Mechanisms
In LLLT, during a process called photodynamic inactivation, a blue light mask is used to kill bacteria. The absorption event occurs in molecules called porphyrins, which are located on the cell membrane. Photosensitization occurs after the appropriate energy is delivered.
During photosensitization, the membrane-bound porphyrin molecules absorb the blue light photons, then generate singlet oxygen radicals that damage or disrupt the cell wall of a variety of Gram-positive bacteria, leading to cell death.
The relatively weak defense mechanisms against singlet oxygen-induced damage contribute to the high efficiency of this treatment option in patients with staphylococcal blepharitis and hordeolum.6
Aesthetics
The LLLT amber light mask is primarily used to treat skin conditions. Results of amber light exposure include improved skin hydration, increased collagen production, increased lymphatic drainage, reduced erythema, decreased melanin synthesis, and decreased metalloproteinase activity.7 Although it is the least used of the three LLLT colors, I use the amber mask for patients with facial erythema, facial edema, and rosacea.
HOW IT WORKS
LLLT is accomplished by using a biostimulation device that incorporates light-emitting diodes (LEDs) or low-powered lasers to deliver excitation energy to the target tissues. In our practice, we use the Epi-C Plus (Espansione Marketing) device, which features a light modulation mask embedded with LED lights (Figure 1).
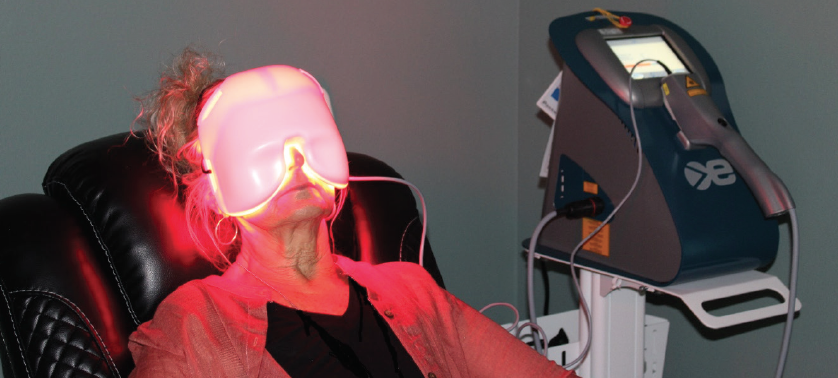 Figure 1. Patient receiving LLLT with a red application mask.
Figure 1. Patient receiving LLLT with a red application mask.
Three mask models are available with the Epi-C Plus (Figure 2); each emits a different color to initiate different biochemical mechanisms within tissues. The mask fits over the patient’s face, covering the forehead and upper facial areas and delivering energy directly over the upper and lower eyelids. Cellular photoactivation can usually be achieved with a 12-to-15-minute application of light.8
During and immediately after the therapy session, the absorption of the light produces a warming effect, causing the patient’s face to feel warm. It should not feel hot, and there should be no discomfort during or after the procedure. There is a low risk of serious adverse effects, and we have successfully treated several patients as young as 3 years old.
A small number of my patients have noticed minor adverse events during or after a therapy session, such as eyelid or facial pain that requires cessation of the therapy, a feeling of excessive heat on the facial areas requiring cessation of therapy, temporary reddening of the skin following a therapy session, and ocular surface irritation or ocular surface inflammation following a therapy session.
PERSONAL EXPERIENCE WITH LLLT
In my clinical experience, the eyelid conditions most responsive to LLLT as monotherapy are meibomian gland dysfunction, staphylococcal blepharitis, chalazion, and hordeolum. We have had exceptional success achieving good outcomes with LLLT as the primary treatment option in these patient populations.
Since adopting this technology, I no longer treat patients with inflammatory eyelid disease using conventional treatments (eg, eyelid scrubs, warm compresses, artificial tears) following a stepwise approach. Adding medications such as steroids, antibiotics, and cyclosporine remains a valuable treatment option, but these are no longer my go-to first-line treatments.
Based on outstanding clinical outcomes after almost 1 year of using this technology, we now offer LLLT as part of our initial treatment program to most of our patients with inflammatory eyelid disease.
A WORTHWHILE INVESTMENT
In my 38 years of practice, LLLT has been one of the most innovative and exciting technologies we have used. The primary purpose of LLLT is to improve cell metabolism and reduce inflammation, and we achieve these goals in most patients. After treatment, patients generally look better and see better, and most report fewer dry eye symptoms and use eye drops less frequently. Based on our practice’s success, I highly recommend adding this technology to your practice.
- 1. Hoon K, Hong-Bee K, Jae-Hwi S, Hongbin K, Kyong Jin C. Effect of 808-nm laser photobiomodulation treatment in blepharitis rat model. Cornea. 2021;40(3):358-363.
- 2. Hamblin MR. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophys. 2017;4(3):337-361.
- 3. Hamblin MR. Mechanisms and mitochondrial redox signaling in photobiomodulation. Photochem Photobiol. 2018;94(2):199-212.
- 4. Poyton, RO, Ball KA. Therapeutic photobiomodulation: nitric oxide and a novel function of mitochondrial cytochrome c oxidase. Discov Med. 2011;11(57):154-159.
- 5. Brown G. Nitric oxide and mitochondrial respiration. Biochim Biophys Acta. 1999;1411(2-3):351-369.
- 6. Sulek A, Pucelik B, Kobielusz M, et al. Photodynamic inactivation of bacteria with porphyrin derivatives: effect of charge, lipophilicity, ROS generation and cellular uptake on their biological activity in vitro. Int J Mol Sci. 2020;21(22):8716.
- 7. Menezes PFC, Urbaczek AC, Matta RFD, Bagnato VS. Photobiomodulation using amber light and infrared laser to control the pigmentation and flaccidity from skin. J Aesthet Reconstr Surg. 2020;6(2):8.
- 8. Stonecipher K, Abell TG, Chotiner E, Potvin R. Combined low level light therapy and intense pulsed light therapy for the treatment of meibomian gland dysfunction. Clin Ophthalmol. 2019;13:993-999.
Craig Thomas, OD
Optometrist, First Eye Care, Dallas thpckc@yahoo.com Financial disclosure: Consultant (Konan Medical)
